Introduction
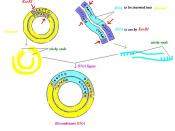
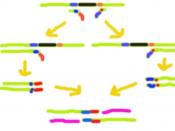
Polymerase chain reaction (PCR), can be used to amplify rare specific DNA sequences into many billions of molecules when the ends of the sequence are known. The method of amplifying rare sequences from a mixture has numerous applications in basic research, human genetics testing, and forensics. In one application, small samples of DNA, such as those found in a strand of hair at a crime scene, can produce sufficient copies to carry out forensic tests. In PCR Genomic DNA is digested into large fragments using a restriction enzyme and then is heat-denatured into single strands. Two synthetic oligonucleotides complementary to the 3' ends of the target DNA segment of interest are added in great excess to the denatured DNA, and the temperature is lowered to 50-60o C. The genomic DNA remains denatured, because the complementary strands are at too low a concentration to encounter each other during the period of incubation, but the specific oligonucleotides, which are at a very high concentration, hybridize with their complementary sequences in the genomic DNA.
The hybridized oligonucleotides then serve as primers for DNA chain synthesis, which begins upon addition of a supply of deoxynucleotides and a temperature-resistant DNA polymerase such as that from Thermus aquaticus (a bacterium that lives in hot springs). The Taq polymerase can extend the primers at temperatures up to 72oC. When synthesis is complete, the whole mixture is heated further (to 95oC) to melt the newly formed DNA duplexes. When the temperature is lowered again, another round of synthesis takes place because excess primer is still present. Repeated cycles of synthesis (cooling) and melting (heating) quickly amplify the sequence of interest. At each round, the number of copies of the sequence between the primer sites is doubled; therefore, the desired sequence increases exponentially (2, 16).
Polymerase Chain...

Not fun to read
this really wasnt the most fun paper to read, but it really wasnt that bad. A lot of good info.
8 out of 10 people found this comment useful.