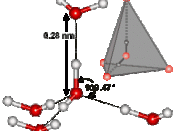
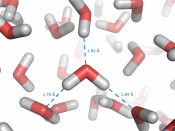
Water behaves differently than almost all other liquids because it expands when it is frozen. As a liquid is cooled down the molecules move more slowly allowing the attractive forces between them to become stronger. Eventually, these forces become strong enough to hold the molecules in a fixed position (they can still vibrate but not change their relative positions) and the liquid becomes a solid. The molecules in the solid have reached an optimum 'closeness' to each other and the solid usually has a very ordered arrangement of molecules in what is known as a crystal lattice. Water is exceptional because it can maximize the attractive forces between its molecules with an arrangement where each water molecule is surrounded by only four other water molecules (If you think of a molecule as spherical - like an orange - you will realize that you could get as many as 12 molecules around it if getting as close together as possible was all that was required! - Just a little note.
;). In many solids this close packing is what happens - but water has an exceptionally strong type of attraction between the molecules (known as hydrogen bonding) and each hydrogen bond is at least 10 times stronger than the other types of attractive forces that hold molecules together. The maximum number of hydrogen bonds a water molecule can form is 4 and in ice each water molecule is doing just that. Water expands to around 9-10% its original volume. So the attractive forces are maximized even though the molecules are forming a very open lattice structure, causing the water to expand and form a block of ice.
Very Little Info
Detailed but not much to it....
8 out of 8 people found this comment useful.