What is radioactivity?
The atoms making up matter are generally stable, but some of them are spontaneously transformed by emitting radiations which release energy. This is called radioactivity.
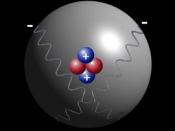
Atoms: In nature, matter - whether water, gas, rocks, living beings - consists of molecules, which are combinations of atoms. Atoms include a positively charged nucleus, around which move negatively charged electrons. The atom is neutral.
The nucleus of the atom includes positively charged protons as well as neutrons. It is this nucleus that is transformed when a radiation is emitted by radioactivity.
Protons and neutrons are, in turn, composed of quarks.
Isotopes
All atoms with nuclei having the same number of protons form a chemical element. Having the same number of protons, they have the same number of electrons, hence the same chemical properties. When they have different numbers of neutrons, they are called "isotopes". Each isotope of a given element is designated by the total number of its nucleons, i.e.
protons plus neutrons.
For instance, Uranium-238 and Uranium-235 both have 92 electrons. Their nuclei have 92 protons. Isotope 238 has 146 neutrons, and Uranium-235 has 143 neutrons.
Radiations from radioactivity
There are three types of radiations corresponding to three types of radioactivity.
Alpha radioactivity corresponds to the emission of a helium nucleus, a particularly stable structure consisting of two protons and two neutrons, called an a particle.
Beta radioactivity corresponds to the transformation, in the nucleus:
-Either of a neutron into a proton, beta- radioactivity, characterized by the emission of an electron e-
- Or of a proton into a neutron, beta+ radioactivity, characterized by the emission of an anti-electron or positron e+. It only appears in artificial radioactive nuclei produced by nuclear reactions.
Gamma radioactivity, unlike the other...


Work cited..
There is no bibliography...
0 out of 0 people found this comment useful.