HYDROGEN BONDING:
Hydrogen bonding is a bonding type consisting of dipole and dispersion forces. A hydrogen bond is the attractive force between a hydrogen atom attached to a molecule and an atom of a different molecule.
According to the Pauling scale of electro-negativities of the elements, it can be viewed that the three most electronegative elements in the periodical table are nitrogen, oxygen and fluorine. These are also called heteroatoms. The heteroatoms have a partial negative charge while hydrogen has a partial positive charge.
Hydrogen bonding is generally stronger than most bonds bar covalent bonds.
Water:
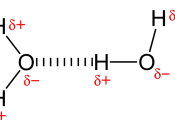
Bonding within water molecules occur between oxygen and hydrogen. These covalent bonds mean that the electrons are shared between the oxygen and hydrogen atoms to create a complete valence shell. However, electrons tend to spend more time around the oxygen atom due to its higher electro-negativity. This creates a partial positive charge around the hydrogen atoms, and a partial negative charge around the oxygen atom.
When other water molecules are present, the negatively charged end of one molecule will be attracted to the positively charged end of the other molecule, as shown below:
Hydrogen bonding is not a type of intramolecular force. Eg. Ionic, metallic and covalent bonding, it is an intermolecular force. An intramolecular force is one that exists between the atoms in a molecule. With water, it is the covalent bond present between the oxygen and two hydrogen atoms being intramolecular and the force that exists between neighboring molecules being intermolecular. Eg. Dispersion forces, dipole-dipole bonds. It is also known that intramolecular forces are much stronger than intermolecular forces. With relation to strength, the covalent bond in water is approximately 25 times the strength of the intermolecular hydrogen bond caused by di-pole attraction.
One type of intermolecular force is dipole-dipole...

OKAY
Needs sources.
Concise. BUT Plagiarism is obviously present. Aren't you gonna credit people???? hmmm??
Hydrogen bonding? something to think about isn't it??
0 out of 0 people found this comment useful.