Coursework: To investigate the effect of temperature changes to the activity of Enzyme Catalyses.
By Marcus
Final update: 26/11/03
Apparatus needed:
Boiling tubes
Beakers
Bung with a delivery tube
Thermometer
5ml syringe (To collect potato)
10ml syringe (To measure the amount of solution)
Diced potato
Bunsen burner (can be replaced by thermosactic water bath if possible)
Ice
Glass rod
Hydrogen peroxide
pH 7 buffer
Variable and invariable factors:
ÃÂpH of enzyme: This can be done by the addition of pH7 buffer.
ÃÂConcentration of enzyme: This can be done by using hydrogen peroxide from the same source every time.
ÃÂThe time for reaction must be the same each time
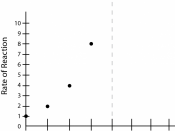
ÃÂSince the effect of changes in temperature to the enzyme activity is investigated in this experiment, all factors, except the temperature, has to be constant. I choose to repeat the experiment several times with a different temperature of the substrate and the enzyme each time.
The temperature chosen are 10âÂÂ,20âÂÂ,
30âÂÂ,40âÂÂ,50âÂÂ,60âÂÂ.
The process:
1.First or all, a boiling tube is used to collect 5ml of diced potato with the 5ml syringe.
2.Add 10ml of pH7 buffer to the boiling tube to make sure pH is constant.
3.Pour 10ml of hydrogen peroxide into another boiling tube.
4.Place both boiling tube in a water bath, add heat to the water bath until it reaches 80 âÂÂ. (For 10âÂÂ. And 20âÂÂ., which is probably lower than room temperature, ice is added to the water bath instead of heating.).
Or
If thermostatic water bath is available, the temperature can be changed by setting the thermostatic water bath. When the temperature is reached, take both boiling tubes out of the thermostatic water bath and continue the experiment.
5. Take both boiling tube out of the water bath, make sure that their temperature are...