The majority of the reactions that occur in living organisms are enzyme-controlled. Without them, the rate of the reactions would be so slow as to cause serious, if not fatal, damage. Without enzymes toxins would soon build up and the supply of respiratory substrate would decrease. Enzymes are proteins and thus have a specific shape. They are therefore specific in the reactions that they catalyse - one enzyme will react with molecules of one substrate. The site of the reaction occurs in an area on the surface of the protein called the active site. Since the active site for all molecules of one enzyme will be made up of the same arrangement of amino acids, it has a highly specific shape. Generally, there is only one active site on each enzyme molecule and only one type of substrate molecule will fit into it. Chymotrypsin and trypsin both catalyse the hydrolysis of peptide bonds but due to their shapes, the active site of chymotrypsin only splits bonds after an aromatic amino acid (one containing a ring of atoms) whereas trypsin only splits bonds after a basic or straight chain amino acid.
This specificity leads to the lock and key hypothesis. However, it has been discovered that competitors for an active site (similar in shape to the substrate) could fit even though they are larger than the substrate. This means that the substrate and active site are a little flexible. This has lead to the induced fit model.
Induced fit model
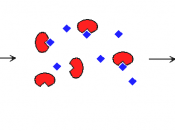
When the enzyme and substrate form a complex, structural changes occur so that the active site fits precisely around the substrate (the substrate induces the active site to change shape). The reaction will take place and the product, being a different shape to the substrate, moves away from the active...

Opinion
A very good essay but it lacks proper references.
0 out of 0 people found this comment useful.