The first thing water reminds us of is the clear liquid in our water bottles. However, water is more than a normal substance. Water is the beginning and the continual of life on earth. Because of its many unique properties, water was able to start life on our planet.
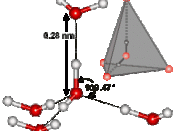
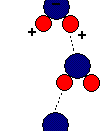
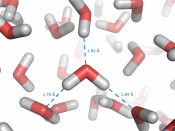
The simple structure of H2O is the source of all water's properties. With two hydrogen atoms sticking to an oxygen atom in a tetrahedron shape, water is considered as a polar molecule. Because oxygen is very electronegative, it pulls hydrogen's electrons towards it, causing oxygen to become partial negative. The hydrogen atoms then will have fewer electrons towards its own nucleus, therefore making it partial positive. Since positive charge and negative charge attracts each other, the molecular formation of H2O will result hydrogen bonding.
Cohesion is an important property of water that greatly helps organisms. One of the significant contributions is the transportation of water in plants against gravity.
When water evaporates from a leaf, other water molecules from further down the vessel will be tugged up due to the hydrogen bonding. The way water molecules attach to each other is called cohesion. Adhesion, the clinging of different substances, also plays a role. Water can hold on to the walls of the vessels to oppose gravity. Because of cohesion and adhesion, water became the prime transportation in the ecological society.
Air temperature can be stabilized by water because water can absorb and release heat. When the temperature is high, water absorbs heat to break the hydrogen bonds; thus, cooling the hot air. When the hydrogen bonds form, heat is released, causing the surrounding air to warm up. This property of water has many advantages because it keeps Earth's temperature shifts within limits that permit life. Also, organisms that are...