DESCRIBE AND EXPLAIN THE STRUCTURE AND FUCTION OF CARBOHYDRATES
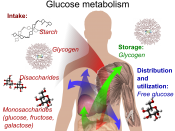
Carbohydrates are part of what we call macronutrients. In our body, as well as proteins and fats, macronutrients are needed in large amounts. Carbohydrates can be monosaccharides (sugars), disaccharides, oligosaccharides and polysaccharides (starch). Carbohydrates have the general formula of Cx(H2O)y- they only contain carbon, hydrogen and oxygen- making them organic compounds.
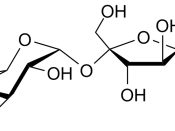
Sugar is a monosaccharide and is very sweet; it is made of alpha glucose monomers. If two of these were to polymerise in a condensation reaction, it would result in a disaccharide. The disaccharide formed would be maltose as it's made from an alpha glucose plus an alpha glucose. The bond formed between the monomers would be a 1,4 - glycosidic bond. Water can be added to polysaccharides to hydrolyse them into smaller components- this is what occurs in our digestive system during the digestion of food molecules, we also use enzymes in this process.
Starch is a polysaccaride - a polymer of monosaccharides that held together by 1,4 - glycosidic bonds. Starch is found in plants is naturally as amylopectin and amylose. Cellulose is made from microfibrils made from cellobiose (a disaccharide) made from beta glucose monomers. Cellulose is based on a rigid, layed structure with many hydrogen cross-linkages between adjacent chains of molecules. Many of these hydrogen bonds collectively provide cellulose with mechanical strength suited to its function. Examples of disaccharides include sucrose, maltose and lactose. They are made from monomer monosaccharides such as glucose, fructose, and galactose.
Starch is a storage molecule and it can store large amounts of energy. Starch is also insoluble so it doesn't affect osmotic concentrations and turgidity and is therefore stored freely in parts of the plant. Starch is very compact and so large amounts of it can be stored for future...