ALKANES
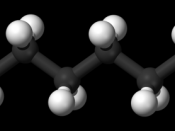
The alkanes are the simplest form of organic compounds. They are made up of
only Carbon atoms and Hydrogen atoms. All of the bonds are single and the number
of hydrogen atoms versus carbon atoms follows this formula: CnH2n+2 Alkanes are all
non-polar molecules so they aren't soluble in water. Here are some more facts.
-Referred to as 'Saturated'
-They have
-low densities
-low melting points
-low boiling points
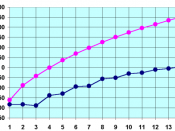
-Refer to 'Slide 29' sheet
We couldn't find any information on who discovered them. Or on the what,
where, or when. However the first alkane that was discovered was probably methane.
Because, of course, this is the gas that cows belch.
The journal of toxology report that a 15 year old boy was stricken with
hemiparesis 'resulting from acute intoxication following inhalation of butane gas.'
Hemiparesis is when half of a person's body is paralyzed. Through reactions alkanes
can be transformed into chloroform.
This has been shown to accumulate in lungs of
swimmers after they swim for extended periods of time.
As mentioned above chloroform can be produced which can be used for
anesthesia. Also dichloromethane, or paint stripper and 1,2-dichloroethane which is a
dry cleaning fluid. Here is a sample reaction where a halogen replaces a hydrogen.
CH4(g) + C12(g) ----> CH3Cl(g) + HCl(g)
There are many uses for alkanes, for instance: Propane is used in gas grills,
butane is used in cigarette lighters, through various reactions scientists can make paint
stripper, anesthesia or dry cleaning fluid. The Pentanes and Hexanes are also highly
flammable and make really cool explosions. Heptane, octane and nonane make up
gasoline. The 'Octane Scale' on gas pumps uses a system which rates n-heptane at a
0 and isooctane at 100.
Currently propane gas is being studied to use it as a fuel for more...