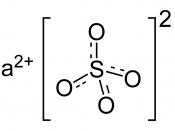
The element Barium has many modern uses and has an impact on the world today. It is used in many things that we see and use everyday and the world would be changed without it in our lives. The element alone however does not have many uses, but when combined with other elements it becomes quite useful. Located in the alkaline earth metal group, Barium has an atomic number of 56 and has an atomic mass of 137.327. The mass is made up of 56 protons and 81 neutrons. There are 6 energy levels of electrons that total 56 and have the S, P, and D orbitals. The electron configuration of Barium is 1s2 2s2p6 3s2p6d10 4s2p6d10 5s2p6 6s2 and is a metallic element that is a soft powder and is silvery white like lead when pure. It also oxidizes very easy and it can be decomposed with water or alcohol.
The density Barium is 3.51 g/cm3 when at 293K and it has a melting point at 998.2 K and boils at 2123 K.
Barium was discovered and recognized as an element by Sir Humphrey Davy in 1808 when he also discovered magnesium, calcium, and strontium. The first person who actually discovered it was Steele in 1774 when he observed a new element in Bononian phosphorus and separated it from the original element, but Davy recognized it as an element. Humphrey Davy was born on December 17, 1778 in Penzance, Cornwall, England and he received his education in Penzance and in Truro. After his father died in 1794 he became an apprentice to a surgeon-apothecary and began it study electrochemistry. Barium got its name from the Greek word barys which means heavy because barium is a heavy element. The actually discovery of the element was in 1774 by Steele and...