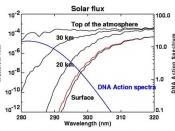
Ozone is a layer of our stratosphere that protects all life on Earth by controlling the temperature of the atmosphere. The ozone layer absorbs the ultraviolet (UV) rays (radiation) of our sun and without this, the temperature of our atmosphere would rise making it impossible for any life to exist on this planet.
This warm layer of modified oxygen exists in our upper stratosphere. Ozone envelops the earth in a protective layer from six to thirty miles above the earth's surface. It is explosive and toxic in high concentrations, but is virtually nonexistent at the earth's surface simply because it decomposes on contact with solid matter.
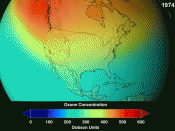
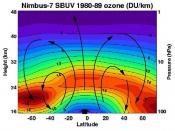
Its concentration is greatest at about 12 to 18 miles above the earth and is only about one ten-thousandth of one percent of our atmosphere. At sea level temperature and pressure it is barely one tenth of an inch thick...pretty meager protection; or so it seems.
In parts of the world where the ozone is the thinnest, we see more cases of sunburn, skin cancer, skin sensitivities, cataracts and cataract blindness. This is evident in people who live at high altitudes.
The oxygen we breathe is made up of molecules with two atoms. Ozone is created when ultraviolet rays split a normal, two-atom oxygen molecule. Each of those atoms then attaches itself to another normal, two-atom molecule, making a three-atom molecule. Ozone is made up of these three-atom molecules.
You can see then that our protective ozone layer is created by UV rays and absorbs UV rays. The three-atom molecules are also destroyed by the UV rays; the UV rays splitting off the extra atom. At the top of the ozone layer this activity is generally in balance. At the lower levels the UV rays have been absorbed by this process and the transformation...